
As the world struggles to cut emissions, attention has turned to carbon capture as part of the climate solution – plucking carbon from the air and locking it away long term. One novel approach is to increase ocean alkalinity, which forces it to draw down additional carbon dioxide from the atmosphere. This week, a US team revealed promising provisional results from an experiment of ocean alkalinity enhancement (OAE) in an open marine environment. They say they quantified the carbon uptake while observing minimal impact on the ecosystem.
The ocean is the largest carbon sink on Earth, holding about 38,000GT of carbon. That’s 40 times more than in the atmosphere and 15–20 times more than the carbon stored in all the world’s land plants and soils.
>> In Other News: RNG Continues to Lead as the Easy-to-switch Clean Fuel for Multi-sector Fleets Signing New Agreements With Clean Energy
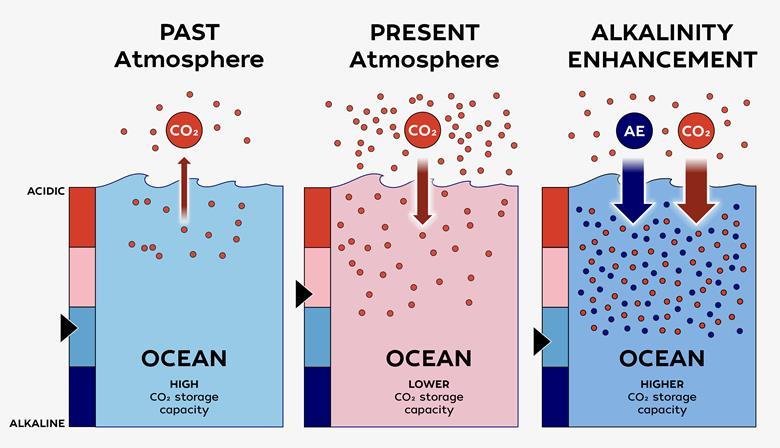
Seawater is already naturally alkaline because it contains dissolved minerals and ions – released from rocks, shells and sediments – that can neutralise acids. This alkalinity causes the ocean to draw down carbon dioxide from the atmosphere, where it forms carbonic acid, which then dissociates into bicarbonate and hydrogen ions.
Over time, however, as additional carbon dioxide dissolves into the seas their pH falls. As the oceans’ pH drops, their capacity to absorb and store additional carbon dioxide declines, weakening one of Earth’s most important natural carbon sinks.

The concept of OAE is simple: add naturally occurring and synthetic alkaline materials to the ocean to make it more alkaline, forcing it to draw down more carbon. ‘Alkalinity enhancement’ is already a restoration tool for lakes and rivers affected by acid rain. But reducing the acidity of the ocean has wider-ranging ecological implications, and it’s much harder to track whether it’s actually working.
That is where the Loc-Ness project comes in, run by the Woods Hole Oceanographic Institution. This interdisciplinary team aims to answer key questions about the effectiveness and impacts of OAE. In August 2025, they ran a six-day field trial in the Gulf of Maine, off the Massachusetts coast.
First, they dispersed 65,000 litres of sodium hydroxide over 1km2. This was accompanied by 760 litres of fluorescent rhodamine dye that was used to monitor how the sodium hydroxide was dispersing throughout a 3600km2 zone.
This test zone was monitored using three vessels and four autonomous underwater vehicles (AUVs). A suite of sensors tracked metrics like water pH and the partial pressure of carbon dioxide. Up above, NASA’s Pace satellite provided a bird’s-eye view of how the chemicals were dispersing.
Presenting their provisional findings at the Ocean Sciences Meeting 2026 in Glasgow, the scientists say an additional 2–10 tonnes of atmospheric carbon dioxide were drawn into the ocean during the four-day monitoring period. Their modelling suggests it could rise to 50 tonnes over the course of a year.
‘These early results demonstrate that small-scale OAE deployments can be engineered, tracked and monitored with high precision,’ says Adam Subhas, a Woods Hole marine chemist and Loc-Ness principal investigator.
The team found no measurable impact on the ecosystem. For instance, no reductions in phytoplankton and zooplankton, and no impacts on organisms higher up the marine food chain. ‘It was really important for us to measure the fish and lobster larvae, after concerns of local fishermen,’ says Rachel Davitt from Rutgers University.

AUVs remain at the test site to track the physical, chemical and ecological impacts over the longer term. To scale up the technology, its developers need to understand how much carbon is truly being offset, as well as the energy needs for chemical production, distribution and measurement.
Loc-Ness scientists emphasise they are not a company looking to sell this technology. But the private sector is pushing ahead anyway. Both Microsoft and Google have signed agreements with Ebb Carbon for carbon offsetting through the release of alkaline waters into the ocean from desalination plants in Saudi Arabia.
Jannes Koelling, an oceanographer at the University of Washington who was not involved in this research, agrees this type of experiment is important for risk analysis. But he warns of unforeseen long-term consequences. ‘We have many examples throughout history of humans using some new technology and only decades later finding out that it caused environmental damage – it’s always hard to predict what will happen when we decide to significantly alter natural processes.’
Correction: The number of litres of rhodamine dye added was 760, not 200 as we stated. Changes were also made to paragraphs 4 and 5 on 3 March 2026 to clarify what is going on during the OAE process.
Subscribe to the newsletter
Daily decarbonization data and news delivered to your inbox
Follow the money flow of climate, technology, and energy investments to uncover new opportunities and jobs.
Latest issues
-
Why Boeing is Buying Crushed Rocks and Charcoal
Inside This Issue ✈️ Boeing Buys 20,000-Ton Portfolio of Biochar, ERW Carbon Removals 📄 Carbonaires Launches RFP for Offtake-Backed Financing of High-Integrity Carbon Removal Projects 🍁 Excluded N...
-
The World Just Got Its First Carbon Mine.
Inside This Issue 🍁 Canada Opens World's First Surficial Mineralization Hub in Quebec 🌳 Living Carbon Secures $500m Deal with Octopus Energy Generation to Restore Degraded Land and Remove CO₂ 🗺️ A...
-
$4.7T in Revenue Just Pushed Back on GHG Protocol
Inside This Issue 🚢 MOL And Alt Carbon Deliver Asia's Largest Verified Erw Credit Batch ⚠️ Apple, Amazon, Schneider Electric Warn GHG Protocol That Tougher Scope 2 Reporting Rules Will Slow Corpor...
Company Announcements
-
Celanese and SharpCell Oy Collaborate to Bring Carbon Capture and Utilization to Airlaid Nonwovens
DALLAS--(BUSINESS WIRE)--Celanese Corporation (NYSE: CE), a global specialty materials and chemical company, and SharpCell Oy, a Finnish family-owned company producing high quality airlaid material...
-
UK May Be Sitting on Volcanic Rocks That Can Swallow Industrial Carbon for Decades
Researchers have found that ancient volcanic rocks beneath the UK could permanently lock away between 42 million and 38 billion tons of industrial carbon dioxide (CO2) by turning it into solid mine...
-
New Carbon-Free Hydrogen Fuel Production Method Slashes Temperature Requirement By 900°F
The study confirmed the catalyst remains stable and functional over ten cycles. Researchers identified BNCF100, a perovskite composed of barium, niobium, calcium, and iron. (Representational image...
-
How Rocks Trap CO₂ Faster: Water-Driven Pathway Could Speed Long-Term Carbon Storage
Rocks can bind carbon dioxide—and much faster than previously thought. For a long time, it was assumed that the transformation of CO2 into carbonate rock depends on very slow, time-consuming proces...