
A photocatalyst has been created that can turn plastic waste into hydrogen and value-added chemicals. Moreover, the reaction repurposes car battery acid, creating a synergistic, sustainable strategy to recycle polymers like polyurethane, polyethylene-terephthalate (PET) and nylon.

Source: © Jupiterimages/Getty Images
The acid from old car batteries could still find a use helping to recycle plastics
Solar reforming is an emerging technology to transform waste products, such as plastics, into clean chemicals using sunlight, explains lead author Erwin Reisner from the Yusuf Hamied chemistry department and St John’s College at the University of Cambridge. Until now, breaking polymers into their basic building blocks presented a problem. ‘The depolymerisation process currently relies on unsustainable alkaline conditions or slow enzymatic degradation,’ explains Reisner. However, this new molybdenum–cobalt photocatalyst is stable in acid, which improves the performance over past approaches.
Moreover, it makes waste valorisation versatile. Whereas previous photoreforming focused on PET, this process expands the scope to other condensation polymers, including polyurethane and nylon. ‘Plastics are a precious resource, which contain key building blocks to build molecules,’ says Reisner. ‘We want to unlock this … to access the circular production of clean chemicals for a defossilised industry.’
‘PET, polyurethane and nylon are common plastics in packaging, coatings and textiles, respectively,’ explains Alexandra Barth, an expert in photocatalysis at North Carolina State University in the US. But despite the ubiquity of plastics, approaches to recycling remain underused and plastic pollution is a persistent problem. Nowadays, ‘most material is non-selectively incinerated’, adds Barth. ‘Approaches to activate [plastics] for polymer recycling, chemical manufacturing and fuel production are highly desirable.’
>> In Other News: SunHydrogen Opens European Headquarters as Global Hydrogen Demand Accelerates
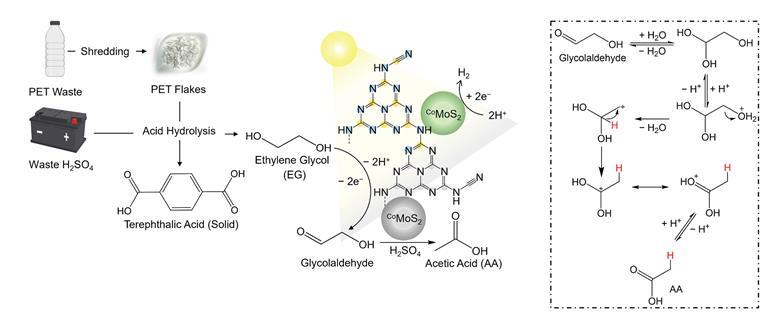
The process starts when battery acid is used to break down plastics such as PET into ethylene glycol. Light-activated oxidation of the ethylene glycol to glycolaldehyde then occurs. The proposed reaction pathway that the molybdenum-cobalt catalyst takes to convert glycolaldehyde into acetic acid is shown on the right
Source: © 2026 Papa K Kwarteng et al/Cell Press
The process starts when battery acid is used to break down plastics such as PET into ethylene glycol. Light-activated oxidation of the ethylene glycol to glycolaldehyde then occurs. The proposed reaction pathway that the molybdenum-cobalt catalyst takes to convert glycolaldehyde into acetic acid is shown on the right
In the study, researchers first use acidic depolymerisation to break plastics down into their constituent monomers. Then, they ‘transform these intermediates into high-value products, including hydrogen and acetic acid’, says Barth. The molybdenum–cobalt catalyst is interesting, because it’s based on abundant materials and driven by visible light, says Barth. ‘Using spent streams of battery acid [also] offers a valuable proof of concept.’ The cobalt catalyst tolerates contaminants and works with recycled sources of sulfuric acid, ‘which is advantageous’, she says.
Nevertheless, the method still has some issues. The process runs with concentrated acid, constant irradiation and temperatures around 140ºC making the process hazardous and energy intensive. Plus, until this approach is prepared to repurpose polyolefins like polyethylene and polypropylene, which make up most of the waste, its impact will be limited.
The team has also carried out technoeconomic analyses demonstrating critical creation of value, particularly with the production of feedstock chemicals. ‘We are currently investigating the use of hydrogen in fuel cells … and [have] established partnerships with plastic recyclers,’ he says. The team is patenting the process and exploring its potential ‘with the tech-transfer arm of the university, Cambridge Enterprise’. Reisner also co-founded Protonera, a University of Cambridge spin-out focused on solar reforming. He’s confident this plastic repurposing process holds ‘significant promise … to create valuable and circular chemicals products’.
References
P K Kwarteng et al, Joule, 2026, 10, 102347 (DOI: 10.1016/j.joule.2026.102347)
Subscribe to the newsletter
Daily decarbonization data and news delivered to your inbox
Follow the money flow of climate, technology, and energy investments to uncover new opportunities and jobs.
Latest issues
-
2.3 Million Tonnes Removed. One Buyer Did Half.
Inside This Issue 🛫 Boeing Backs $10M Quebec SAF Project to Fly by 2027 🏗️ Eni CCUS Holding Expands the Financing Sources for Its Platform of CCS Projects 🍁 GeoRedox and Canada Nickel Launch First...
-
EPA Repeal Rule Heads to White House — Big Shift for Carbon Projects?
Inside This Issue 🏛️ EPA Sends Final Rule to Repeal Power Plant Greenhouse Gas Standards to White House for Review 🌬️ NTT DATA Partners with Climeworks for Carbon Removal 🏭 Honeywell Gives a Shutt...
-
100M Gallons of SAF, No Trucks Required
Inside This Issue ✈️ Pittsburgh Airport Is Building America's First On-Site SAF Plant 📝 Wren's 2026 Request for Proposals 🍁 Canada Expands CCUS Investment Tax Cr to Include Enhanced Oil Recovery 🏭...
Company Announcements
-
CUPERTINO, Calif., May 21, 2026 (GLOBE NEWSWIRE) -- Aemetis, Inc. (NASDAQ: AMTX), a diversified renewable natural gas and biofuels company, announced today that the Capital Programs & Climate F...
-
Initial high-priority drill targets finalized for continuous follow-up program at Lawson Natural Hydrogen Complex to speed up timeline for potential commercialization Genesis Explained: Its “Salt ...
-
Deep Sky and Lufthansa Group Enter Carbon Removal Credit Agreement
Senken supported due diligence and project vetting on a transaction centred on quality, credibility, and long-term delivery. MONTREAL, May 21, 2026 /PRNewswire - Deep Sky has entered into an offta...
-
Hyundai Motor Group Showcases Hydrogen Ecosystem Vision for Europe at World Hydrogen Summit 2026
Hyundai Motor Group showcases its hydrogen ecosystem vision for Europe at the World Hydrogen Summit 2026 in Rotterdam The Group's display of the all-new NEXO and hydrogen fuel cell systems underli...